Roche’s Alzheimer’s Drug Doesn’t Meet Goals in Trials
An Alzheimer’s drug being tested by pharmaceutical company Roche has failed to meet its goals.
In a news release, Roche provided an update on monoclonal antibody gantenerumab, which has been tested in two drug trials, Graduate 1 and Graduate 2. The company said neither trial has seen the drug meet its main goal of slowing clinical decline in the early stages of Alzheimer’s. The findings will be shared later this month at the Clinical Trials on Alzheimer’s Disease (CTAD) Conference.

Dr. Levi Garraway, Roche’s Chief Medical Officer and Head of Global Product Development, says, “We are profoundly grateful to the study participants, their care partners and study sites for their contributions to this research. While the GRADUATE results are not what we hoped, we are proud to have delivered a high quality, clear and comprehensive Alzheimer’s dataset to the field, and we look forward to sharing our learnings with the community as we continue to search for new treatments for this complex disease.”
Gantenerumab is designed to target and bind to forms of beta-amyloid and activate immune cells in the brain to help clear amyloid plaques and stop them from accumulating further. In the trials, which were conducted with nearly 2,000 participants across 30 countries, the drug was tested in those with either mild cognitive impairment due to Alzheimer’s or mild Alzheimer’s.

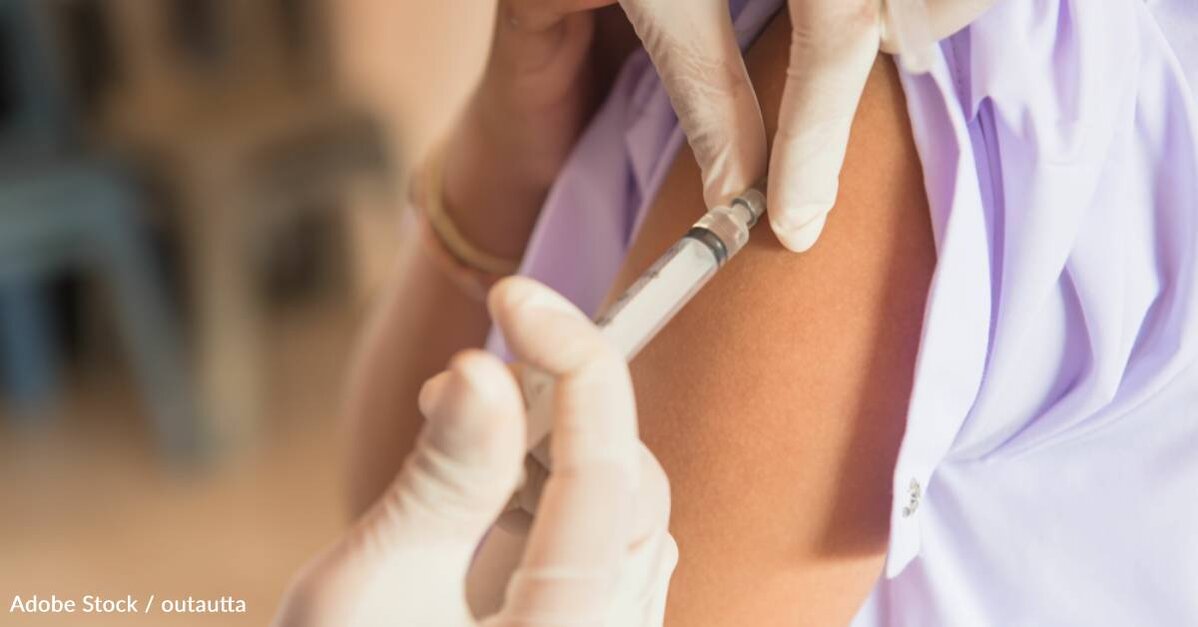
For 27 months, patients were either injected with 510mg of gantenerumab every two weeks, or they received a placebo. Their cognitive health was then tested at 116 weeks using the Clinical Dementia Rating-Sum of Boxes, which looks specifically at memory, orientation, judgment and problem solving, community affairs, home and hobbies, and personal care.
The findings showed that Graduate 1 trial participants who had received gantenerumab had an 8% slowing of decline, compared with placebo, while those in the Graduate 2 trial showed a 6% reduction. However, the company says this isn’t statistically significant.
Going forward, Roche says it is still developing tests to help with early Alzheimer’s diagnosis and is working on other future medications for the disease.
